 Loading
......
Loading
......
PseudoUridine
PseudoUridine, a metabolite found in Escherichia coli (strain K12, MG1655), is a C-glycosyl pyrimidine consisting of uracil with a β-D-ribofuranose residue attached at the 5th position. The C-glycosyl isomer of the nucleoside uridine. It has a role as a fundamental metabolite.
| Name | PseudoUridine |
| CAS | 1445-07-4 |
| Structure |  |
| Molecular Formula | C9H12N2O6 |
| Synonyms | beta-PseudoUridine psi-Uridine 5-(beta-D-Ribofuranosyl)uracil 5-((2S,3R,4S,5R)-3,4-Dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione UNII-7R0R6H6KEG NSC 162405 7R0R6H6KEG |
| Molecular Weight | 244.20 |
| Density | 1.641±0.06 g/cm3 (Predicted) |
| Melting Point | 222℃ |
| Acidity Factor | 8.52±0.10 (Predicted) |
| Storage Conditions | Inert atmosphere, room temperature |
| Physical Property | The most abundant modified nucleotide in non-coding RNA enhances the function of tRNA and rRNA by stabilizing the RNA structure. |
PseudoUridine Modifications
PseudoUridine modifications are the most abundant RNA modifications and generally arise from the isomerization of uridine. PseudoUridine modifications can be achieved by two different mechanisms, RNA-dependent and RNA-independent pseudouridylation.
- The RNA-dependent mechanism relies on the RNA-protein complexes (DKC1, Nhp2, Nop10 and Gar1) of H/ACA snoRNPs. In H/ACA, ncRNA is responsible for substrate recognition through complementary base-pairing interactions with RNA substrates and DKC1 provides the catalytic activity.
- RNA-independent pseudoUridine chemistry is catalyzed by a single enzyme, pseudoUridine synthase (PUS), which performs both substrate recognition and catalysis in the absence of the RNA template strand. rules for RNA substrate recognition by RNA-independent pseudoUridine synthase have only been elucidated in a few cases, e.g. pseudoUridine synthase has a rather stringent substrate specificity and can only modify only one site in the cellular RNA (e.g. tRNAs).
rRNA
Ribosomal RNA, rRNA, is the most abundant type of RNA in cells and has the largest relative molecular mass of the three types of RNA (tRNA, mRNA and rRNA). It combines with proteins to form ribosomes, whose function is synthesizing amino acids into peptide chains under the direction of mRNA (The peptide chains are twisted and folded into proteins under the action of the endoplasmic reticulum and Golgi apparatus, which are completed in prokaryotes in the cytoplasm). rRNA accounts for about 82% of all RNA.
rRNA does not perform its function when it exists alone, it combines with various proteins to form ribosomes, which act as "assembly machines" for protein biosynthesis. The molecular weight of rRNA is relatively large and the structure is quite complex. Although the primary structure of many rRNA molecules has been measured, the research on its secondary and tertiary structures and their functions need further in-depth research.
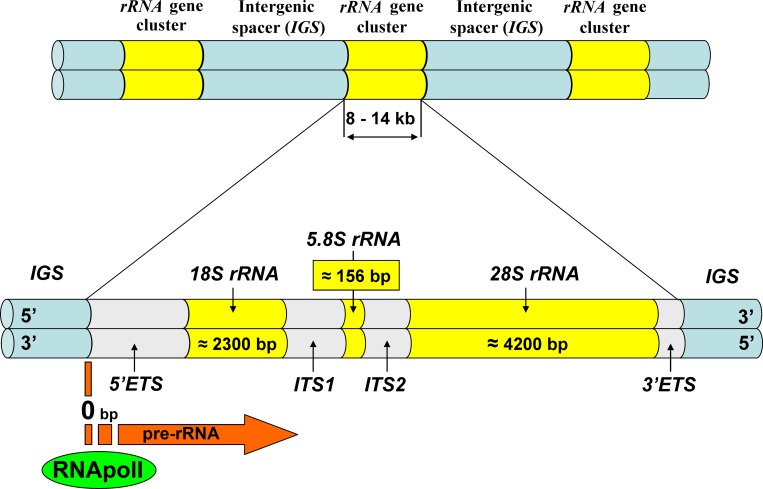 Fig. 1 Ribosomal DNA structure (Dyomin AG, 2016)
Fig. 1 Ribosomal DNA structure (Dyomin AG, 2016)
PseudoUridine Modifications in rRNA
PseudoUridine modifications in rRNA are commonly found in the ribosomal large subunit (RLS) and ribosomal small subunit (RSS) of rRNA, and are mainly clustered in functionally important regions, such as the peptidyl transferase center (PTC), decoding center and A-site finger region (ASF). Its distribution suggests that pseudoUridine modifications can affect rRNA function. PseudoUridine modifications in these regions may affect rRNA folding, ribosome assembly and maintenance of the corresponding high-level structure. When the pseudoUridine modifications in the PTC region of the yeast cells were absent by knocking out the snoRNA associated with this region, although a single snoRNA deletion was found to have less effect on growth when all snoRNAs were absent it had a large effect on growth rate. The absence of pseudoUridine in the decoding center causes a slow growth rate, a significant defect in the small subunit RNA and a slow rate of amino acid incorporation.
Reference
- Dyomin AG; et al. Chicken rRNA gene cluster structure. PLoS One. 2016 Jun 14; 11(6): e0157464.